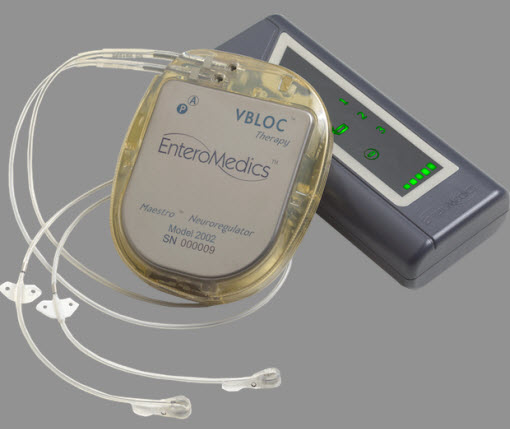
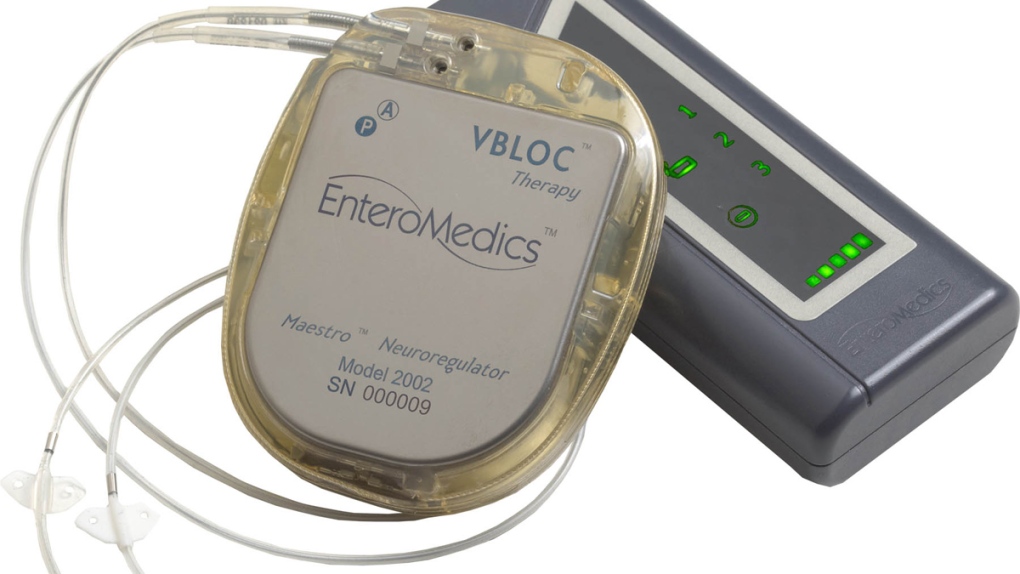
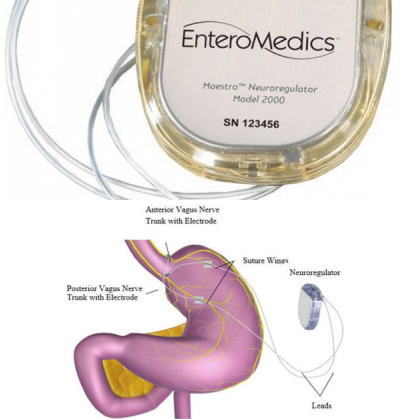
![PDF] Update on bariatric surgical procedures and an introduction to the implantable weight loss device: the Maestro Rechargeable System | Semantic Scholar PDF] Update on bariatric surgical procedures and an introduction to the implantable weight loss device: the Maestro Rechargeable System | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/e6f7c4efe5b13d962ce15bcc46dd35223d2fff59/2-Figure1-1.png)
PDF] Update on bariatric surgical procedures and an introduction to the implantable weight loss device: the Maestro Rechargeable System | Semantic Scholar
The Maestro Rechargeable System - New Weight-Loss Surgical Product Approved By FDA - Clinique Michel Gagner
MC2402 Maestro Rechargeable System Mobile Charger RF Exposure Info RF Exposure Report Enteromedics, .