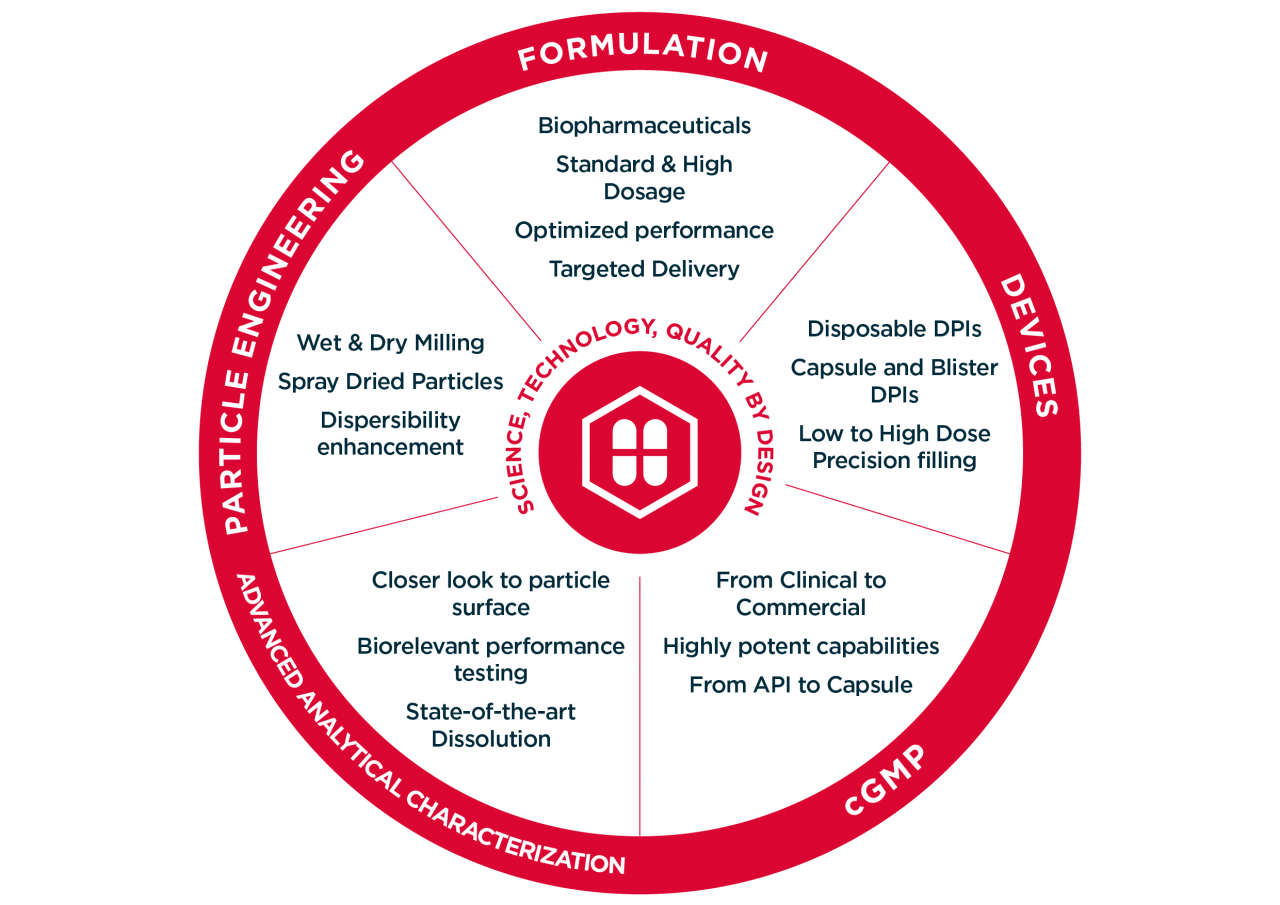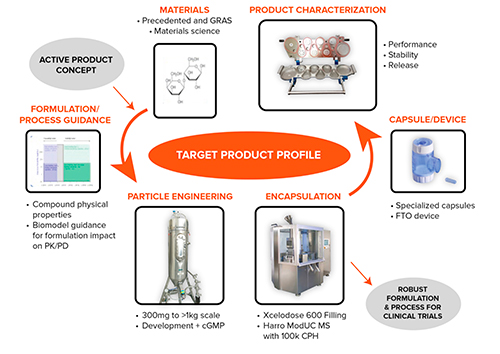
ORALLY INHALED PRODUCTS - Optimizing the Application of In Vitro Test Methods for the Demonstration of Bioequivalence in Orally Inhaled Products

R&Y International on LinkedIn: #inhalation #respiratory #drugdelivery #conference #inhaler #inhalers…

Inhaled formulation and device selection: bridging the gap between preclinical species and first-in-human studies | Therapeutic Delivery
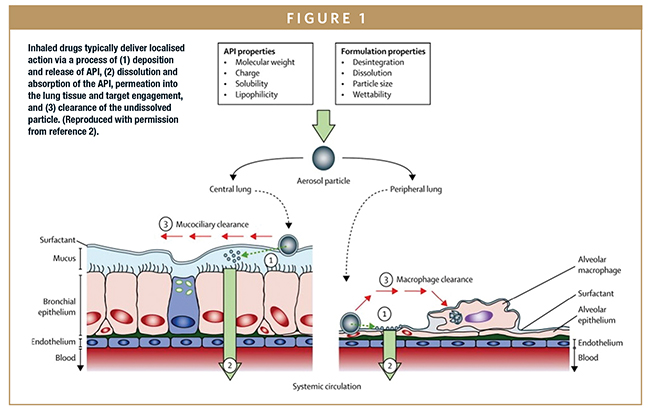
Frontiers | Half a Century of Technological Advances in Pulmonary Drug Delivery: A Personal Perspective

Inhaled Pharmaceutical Product Development Perspectives: Challenges and Opportunities (Emerging Issues in Analytical Chemistry): 9780128122099: Medicine & Health Science Books @ Amazon.com

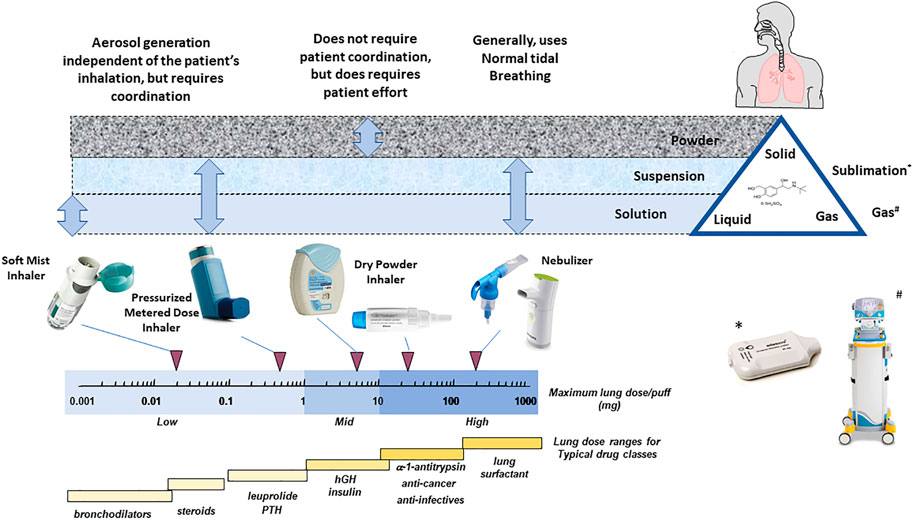
Contemporary Formulation Development for Inhaled Pharmaceuticals - Journal of Pharmaceutical Sciences

iBCS: 2. Mechanistic Modeling of Pulmonary Availability of Inhaled Drugs versus Critical Product Attributes | Molecular Pharmaceutics