EU Medicines Agency on X: "#EMA supports #RareDiseaseDay every day! Our Committee for Orphan Medicinal Products is the only committee fully dedicated to medicines for rare diseases. #ShowYourRare https://t.co/uNumz8tDDX https://t.co/heMlVfyYnm" / X

Frontiers | Licensing of Orphan Medicinal Products—Use of Real-World Data and Other External Data on Efficacy Aspects in Marketing Authorization Applications Concluded at the European Medicines Agency Between 2019 and 2021
Celleron Therapeutics announces EMA COMP Orphan Drug designation for CXD101 in peripheral T-cell lymphoma

GRANDPRIX TV - COMPIEGNE CSIOY AVRIL 2023 - Competition 09 - Prix LA DADA BOX - BLUELOVE DU CANDY - EMA LEGAY
Public summary of opinion on orphan designation Cisplatin for the treatment of malignant mesothelioma
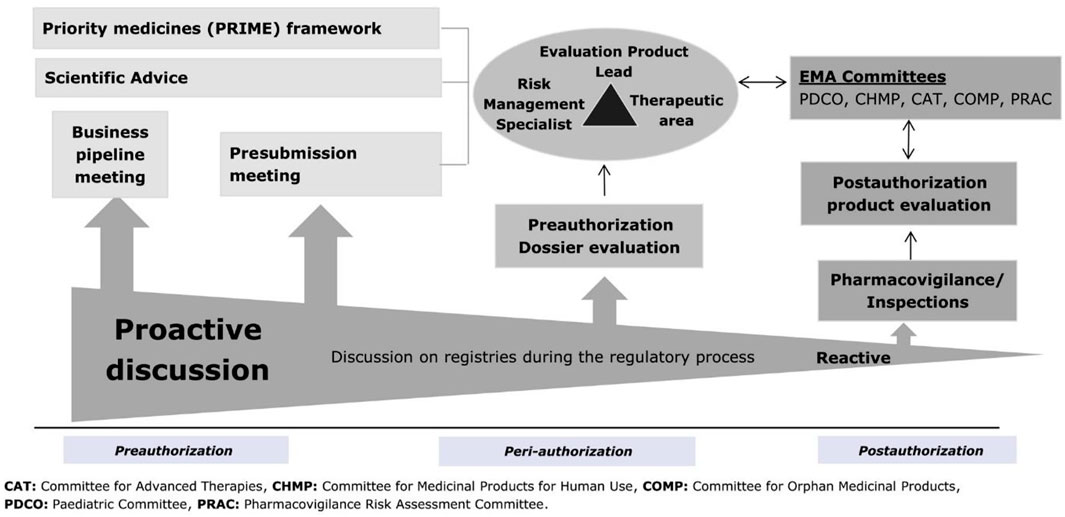
Frontiers | Contribution of patient registries to regulatory decision making on rare diseases medicinal products in Europe

Cantargia receives positive opinion from EMA (COMP) - Nordic Life Science – the leading Nordic life science news service

Use of biomarkers in the context of orphan medicines designation in the European Union – topic of research paper in Clinical medicine. Download scholarly article PDF and read for free on CyberLeninka